 We begin by assuming a Lewis structure model for chemical bonding based on valence shell electron pair sharing and the octet rule.Disputemus 27K views 9 years ago A step-by-step explanation of how to draw the Disulfur Dichlorine Lewis Dot Structure (Monochloramine). Oxygen belongs to group 16 ( or group 6) and has an atomic number of 8, therefore a valency of 6.NH3Cl+ ut chloramine protonato dici potest, phaselus gasi intermedius formatus est cum chloramine, NH2Cl cum protono speciebus donantibus reagit. Nitrogen belongs to group 15 ( or group 5) and has an atomic number of 7, therefore has a valency of 5. A molecule of nitrogen dioxide consists of one nitrogen atom and two atoms of oxygen. Similarly, dsp 3 includes one s, three p, and one d-orbital.Lewis Structure of NO2. For example, sp 2 orbital includes one s and two p orbital. The names of these orbital include all the participating orbital. Hybridization is the process of the intermingling of different orbital of similar energy to form a new orbital. In XeF6 Molecular Geometry, there are eight electrons present in the outer shell of the xenon. The bond angle of xenon hexafluoride is 90° and 72°. A positively charged polyatomic ion of Ammonium or NH4+ comes into existence when an Ammonia atom goes through the process of protonation, that is, it loses … bluueygooey It describes how many lengths angles one atom of xenon hexafluoride is attached to another atom. Media Atom assignans NH3 is the chemical formula of Ammonia. Sed crimen positivum detrahit unum electronicum e 15 so 14 electrons valoris in promptu. Hinc summa electronicorum valentium XV praesto sunt ad educendum NH. Cl dat 7 et N dat 5 electrons valentia 2s, 2p, 3s et 3p. c.Tres H atomi dant 3 electrons valentes. 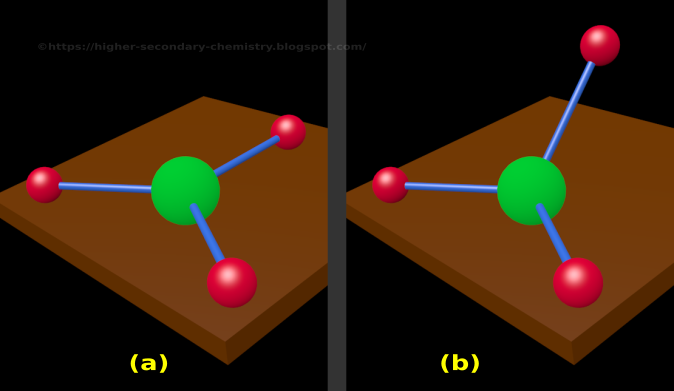
What is the electron-pair geometry about the central atom applying VSEPR theory? Sketch it. Include all the valence electrons and formal charges. 1 watching 2,547 views greyelephant419 Lv1 For the molecular ion SeF 5-, a. Include resonance structures and assign formal charges to each atom, a) CO, b) 0 c) CH,CO (acetate ion) Draw Lewis structures of the following. If there are no lone electron pairs on the central atom, the electron pair and molecular geometries are the same.Ĭlick here to see the various molecular geometries.Ĭhoose the correct molecular geometries for the following molecules or ions below.Nh3cl+ lewis structure Question: Draw Lewis structures of the following. Step 4: The molecular geometry describes the position only of atomic nuclei (not lone electron pairs) of a molecule (or ion). * Lone electron pairs are represented by a line without an atom attached. The table below shows the electron pair geometries for the structures we've been looking at: The regions of electron density will arrange themselves around the central atom so that they are as far apart from each other as possible. Use this number to determine the electron pair geometry.Įach bond (whether it be a single, double or triple bond) and each lone electron pair is a region of electron density around the central atom. Step 3: Add these two numbers together to get the regions of electron density around the central atom. Make sure you understand why they are correct. The correct answers have been entered for you. How many lone electron pairs are on the central atom in each of the following Lewis structures? Step 1: Count the number of lone electron pairs on the central atom. Molecular geometry can be predicted using VSEPR by following a series of steps: In other words, the electrons will try to be as far apart as possible while still bonded to the central atom. The theory says that repulsion among the pairs of electrons on a central atom (whether bonding or non-bonding electron pairs) will control the geometry of the molecule. The valence shell electron-pair repulsion theory (abbreviated VSEPR) is commonly used to predict molecular geometry. Lewis structures are very useful in predicting the geometry of a molecule or ion. Many of the physical and chemical properties of a molecule or ion are determined by its three-dimensional shape (or molecular geometry). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
